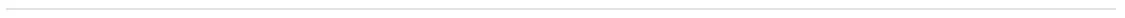EXPAND
Long-term safety of siponimod
Kappos L, et al. Long-term efficacy and safety of siponimod in patients with SPMS: EXPAND extension analysis up to 5 years. Oral presentation at AAN. 2020;S40.003.
- IRs of the most common AEs reported in the core+extension period were consistent with those of the core period. The long-term safety profile of siponimod for up to 5 years remained consistent with that of the core study.
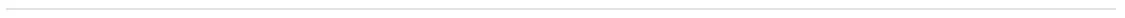
BOLD and EXPAND
Long-term safety of siponimod
Kappos L, et al. Longer-term safety with siponimod treatment in multiple sclerosis: Pooled** analysis of data from the BOLD and EXPAND trials and their extensions. Poster presentation at ECTRIMS. 2018;P911.
- Treatment over 2 years with siponimod 2 mg did not reveal an increase in the incidence of AEs with time, and no new safety findings were observed in the long-term pool versus the controlled pool
- The incidence of VZV infections was higher with siponimod 2 mg versus placebo in the controlled pool, with no further increase in the IR in the long-term pool
- No increase in the overall IR of malignant or unspecified tumours including skin malignancies was observed in the long-term pool versus the siponimod 2 mg and placebo groups of the controlled pool
**The pooled population comprised a controlled pool (all patients who received siponimod 2.0 mg [N=1148] or placebo [N=607] during the core part of the placebo-controlled studies) and a long-term pool (all patients who received at least one dose of siponimod 2.0 or 10.0 mg in the core/extension parts [N=1737])